よむ、つかう、まなぶ。
参考資料13 Vaccines and Related Biological Products Advisory Committee June 7, 2022 Meeting Presentation - FDA Review of Effectiveness and Safety of Novavax COVID-19 Vaccine in Adults > 18 Years of Age - Emergency Use Authorization Request(Vaccines and Related Biological Products Advisory Committee June 7, 2022 Meeting FDA提出資料) (24 ページ)
出典
| 公開元URL | https://www.mhlw.go.jp/stf/shingi2/0000208910_00043.html |
| 出典情報 | 第80回厚生科学審議会予防接種・ワクチン分科会副反応検討部会、令和4年度第5回薬事・食品衛生審議会薬事分科会医薬品等安全対策部会安全対策調査会(合同開催)(6/10)《厚生労働省》 |
ページ画像
ダウンロードした画像を利用する際は「出典情報」を明記してください。
低解像度画像をダウンロード
プレーンテキスト
資料テキストはコンピュータによる自動処理で生成されており、完全に資料と一致しない場合があります。
テキストをコピーしてご利用いただく際は資料と付け合わせてご確認ください。
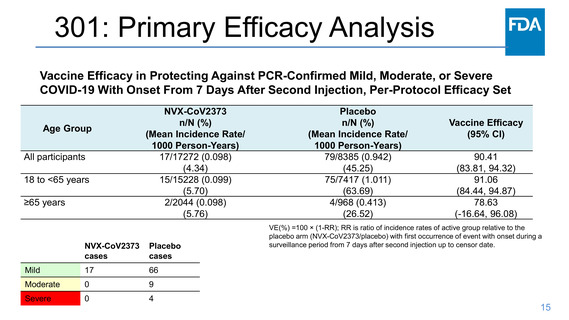
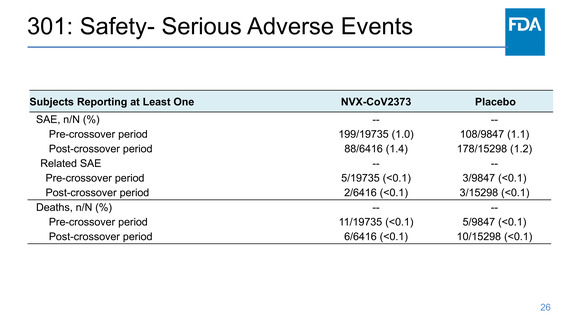
301: Safety- Solicited systemic reactogenicity
Frequency of Solicited Systemic Reactions Within 7 Days After Each
Dose, Safety Analysis Set Participants 18 to <65 Years of Age
NVXCoV2373
Dose 1
N=15884
Joint pain (arthralgia), (%)
-Any (Grade ≥1)
7.9
Grade 1
4.9
Grade 2
2.7
Grade 3
0.3
Grade 4
<0.1
Nausea/vomiting, (%)
-Any (Grade ≥1)
6.7
Grade 1
5.4
Grade 2
1.2
Grade 3
0.1
Grade 4
<0.1
Fever, (%)
-Any (Grade ≥1)
0.4
Grade 1
0.2
Grade 2
0.1
Grade 3
<0.1
Grade 4
<0.1
Solicited Systemic
Reaction
Placebo
Dose 1
N=7868
-6.6
4.1
2.2
0.3
0
-5.9
4.6
1.2
0.1
<0.1
-0.4
0.2
0.1
0.1
<0.1
NVXCoV2373
Dose 2
N=15148
-23.4
9.8
10.9
2.6
<0.1
-12.0
8.6
3.2
0.2
0.1
-6.2
4.1
1.7
0.4
<0.1
Frequency of Solicited Systemic Reactions Within 7 Days After
Each Dose, Safety Analysis Set Participants ≥65 Years of Age
Placebo
Dose 2
N=7361
-6.9
4.3
2.2
0.3
<0.1
-5.7
4.3
1.2
0.1
<0.1
-0.2
0.2
<0.1
<0.1
0
Solicited Systemic
Reaction
Joint pain (arthralgia), (%)
Any (Grade ≥1)
Grade 1
Grade 2
Grade 3
Grade 4
Nausea/vomiting, (%)
Any (Grade ≥1)
Grade 1
Grade 2
Grade 3
Grade 4
Fever, (%)
Any (Grade ≥1)
Grade 1
Grade 2
Grade 3
Grade 4
NVXCoV2373
Dose 1
N=2251
-6.2
3.8
2.2
0.2
0
-3.6
3.1
0.5
0
0
-0.4
0.2
0.1
<0.1
0
Placebo
Dose 1
N=1114
-6.4
3.5
2.5
0.4
0
-2.9
2.3
0.5
0
0
-0.3
0.2
0.1
0
0
NVXCoV2373
Dose 2
N=2048
-13.2
6.9
5.5
0.8
0.1
-5.3
4.1
1.0
0.2
0
-2.0
1.5
0.4
0.1
0
Placebo
Dose 2
N=978
Fever: Grade 1: 38.0 to 38.4°C/100.4 to 101.1°F; Grade 2: 38.5 to 38.9°C/101.2 to 102.0°F; Grade 3: 39.0 to 40°C/102.1 to 104°F; and Grade 4: >40°C/>104°F.
Headache: Grade 1: No interference with activity; Grade 2: Repeated use of nonnarcotic pain reliever >24 hours or some interference with activity; Grade 3: Significant; any use of narcotic pain
reliever or prevents daily activity; and Grade 4: ER visit or hospitalization.
Fatigue/malaise: Grade 1: No interference with activity; Grade 2: Some interference with activity; Grade 3: Significant; prevents daily activity; and Grade 4: ER visit or hospitalization.
Myalgia/arthralgia: Grade 1: No interference with activity; Grade 2: Some interference with activity; Grade 3: Significant; prevents daily activity; and Grade 4: ER visit or hospitalization.
Nausea/vomiting: Grade 1: No interference with activity or 1 to 2 episodes/24 hours; Grade 2: Some interference with activity or >2 episodes/24 hours; Grade 3: Prevents daily activity, requires
outpatient IV hydration; and Grade 4: ER visit or hospitalization for hypotensive shock.
-6.4
3.5
2.8
0.2
0
-3.6
2.9
0.7
0
0
-0.7
0.5
0.1
0.1
0
23
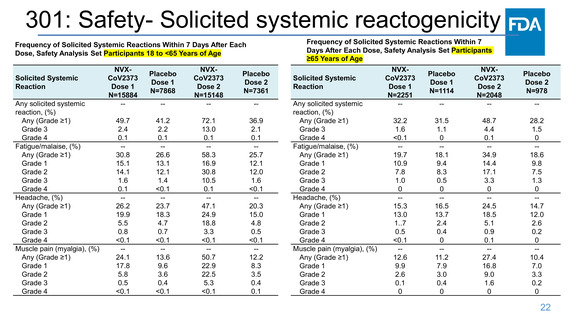
Frequency of Solicited Systemic Reactions Within 7 Days After Each
Dose, Safety Analysis Set Participants 18 to <65 Years of Age
NVXCoV2373
Dose 1
N=15884
Joint pain (arthralgia), (%)
-Any (Grade ≥1)
7.9
Grade 1
4.9
Grade 2
2.7
Grade 3
0.3
Grade 4
<0.1
Nausea/vomiting, (%)
-Any (Grade ≥1)
6.7
Grade 1
5.4
Grade 2
1.2
Grade 3
0.1
Grade 4
<0.1
Fever, (%)
-Any (Grade ≥1)
0.4
Grade 1
0.2
Grade 2
0.1
Grade 3
<0.1
Grade 4
<0.1
Solicited Systemic
Reaction
Placebo
Dose 1
N=7868
-6.6
4.1
2.2
0.3
0
-5.9
4.6
1.2
0.1
<0.1
-0.4
0.2
0.1
0.1
<0.1
NVXCoV2373
Dose 2
N=15148
-23.4
9.8
10.9
2.6
<0.1
-12.0
8.6
3.2
0.2
0.1
-6.2
4.1
1.7
0.4
<0.1
Frequency of Solicited Systemic Reactions Within 7 Days After
Each Dose, Safety Analysis Set Participants ≥65 Years of Age
Placebo
Dose 2
N=7361
-6.9
4.3
2.2
0.3
<0.1
-5.7
4.3
1.2
0.1
<0.1
-0.2
0.2
<0.1
<0.1
0
Solicited Systemic
Reaction
Joint pain (arthralgia), (%)
Any (Grade ≥1)
Grade 1
Grade 2
Grade 3
Grade 4
Nausea/vomiting, (%)
Any (Grade ≥1)
Grade 1
Grade 2
Grade 3
Grade 4
Fever, (%)
Any (Grade ≥1)
Grade 1
Grade 2
Grade 3
Grade 4
NVXCoV2373
Dose 1
N=2251
-6.2
3.8
2.2
0.2
0
-3.6
3.1
0.5
0
0
-0.4
0.2
0.1
<0.1
0
Placebo
Dose 1
N=1114
-6.4
3.5
2.5
0.4
0
-2.9
2.3
0.5
0
0
-0.3
0.2
0.1
0
0
NVXCoV2373
Dose 2
N=2048
-13.2
6.9
5.5
0.8
0.1
-5.3
4.1
1.0
0.2
0
-2.0
1.5
0.4
0.1
0
Placebo
Dose 2
N=978
Fever: Grade 1: 38.0 to 38.4°C/100.4 to 101.1°F; Grade 2: 38.5 to 38.9°C/101.2 to 102.0°F; Grade 3: 39.0 to 40°C/102.1 to 104°F; and Grade 4: >40°C/>104°F.
Headache: Grade 1: No interference with activity; Grade 2: Repeated use of nonnarcotic pain reliever >24 hours or some interference with activity; Grade 3: Significant; any use of narcotic pain
reliever or prevents daily activity; and Grade 4: ER visit or hospitalization.
Fatigue/malaise: Grade 1: No interference with activity; Grade 2: Some interference with activity; Grade 3: Significant; prevents daily activity; and Grade 4: ER visit or hospitalization.
Myalgia/arthralgia: Grade 1: No interference with activity; Grade 2: Some interference with activity; Grade 3: Significant; prevents daily activity; and Grade 4: ER visit or hospitalization.
Nausea/vomiting: Grade 1: No interference with activity or 1 to 2 episodes/24 hours; Grade 2: Some interference with activity or >2 episodes/24 hours; Grade 3: Prevents daily activity, requires
outpatient IV hydration; and Grade 4: ER visit or hospitalization for hypotensive shock.
-6.4
3.5
2.8
0.2
0
-3.6
2.9
0.7
0
0
-0.7
0.5
0.1
0.1
0
23